Macaques do not show signs of pain and poor health as obviously as humans; therefore, regular observation and evaluation of health is essential. Keeping detailed records regarding the external condition of individual animals will aid in the early detection of changes indicating poor health. Score sheets (see the tab below) allow records to be kept consistently across time and between staff (who may interpret clinical signs differently). Clinical examinations involving sedation and removal from the group should always be performed by a qualified veterinarian. Click here for mean measurements of anthropometric parameters (e.g. head, waist, total length and weight) for rhesus macaques (scroll to the end of the document).
Adapted from Tasker 2012; see the tabs above for further information on body condition scoring hair loss, and assessing pain.
Health indicators (bold) and changes indicative of impaired welfare are described below.
Body condition
-
Dramatic weight loss such that ribs, hips and spine become visible: Body condition scores can differ depending on age, growth pattern and reproductive status
-
Dramatic weight gain: Condition scoring of weight may be performed non-invasively through observation, or during clinical examination by palpating the thoracic and lumbar vertebrae to assess whether weight changes exceed healthy limits [1,2].
-
Poor growth: Regular condition scoring and body weight assessment at weaning and in the post-weaning period are especially important to ensure young animals receive adequate nutrition to support growth and normal development [2]. Monitoring of growth is also especially important for macaques on food and fluid control protocols [3]. Growth curves are available in the published literature [3-7].
-
Signs of lameness or imbalance when moving
-
Wounds may indicate: 1) attack from conspecifics due to problems in social environment (e.g. lack of visual barriers and escape routes for subordinates; or 2) self-harm such as self-biting. Evident wounds indicate advanced pathology, which requires immediate intervention.
Pelage
-
Patchy hair loss indicates: 1) an underlying bacterial or parasitic skin infection; 2) self-directed hair plucking due to boredom and stress in an impoverished environment; or 3) hair pulling by conspecifics due to problems in social environment. Novak & Meyer (2009) provide a comprehensive review of possible causes of hair loss.
-
Reduction in shine and quality
-
Changes in colour
Faeces
-
Diarrhoea: One of the most common causes morbidity and mortality in captive populations [8-10]. Diarrhoea may indicate acute stress or underlying illness. Carries risk of dehydration, electrolyte imbalance and weight loss. Post-weaning diarrhoea is predicted by weight at weaning, and also bouts of pre-weaning diarrhoea. These combined factors predict high risk of reduced growth, smaller weight and impaired immune function [2].
Urination
-
Changes include: Excessive urination, lack of urination, or urine drinking
Face
-
Discharge or foam at the mouth, nostrils or eyes
Posture
-
Piloerection or hunched posture: Piloerection indicates arousal and may be caused by aggression in the group or towards staff (see the Postures tab under Behaviour). Hunched unresponsive or avoidant posture may indicate pain or depression (see the Signs of poor welfare tab under Behavioural indicators).
Breathing
-
Heavy fast or laboured
-
Coughing: Like humans, macaques catch colds which they recover from naturally. Veterinary assessment is required for coughs, as this may indicate more serious underlying pathology.
Body condition can be assessed by visual judgement as to the amount of muscle and subcutaneous fat [1,2]. It can be used as guide to assess level of nutrition [1], whether the animal is undergoing adequate growth or experiencing poor health (e.g. chronic diarrhoea) [1], and as a useful parallel measure with body weight. Two scoring methods for adult rhesus macaques have been published [1,11]. The interpretation of the significance of body condition scores should be made in light of the animal’s current age and reproductive status (e.g. juvenile animals tend to be lean and lanky, scoring as underweight or thin; an animal that has just undergone a growth spurt may be evaluated as thin, whereas one whose growth has stabilized may be lean, as muscle mass catches up with previous bone growth) [1]. The scoring system however, needs to be sensitive enough to pick up changes in growth and body weight coinciding with events in the laboratory if it is to be a useful welfare assessment tool.
Condition scoring by palpation
Body condition can also be scored by palpating the monkey over its thoracic and lumbar vertebrae (at the level of the last rib), making a judgement as to the amount of fat and muscle covering the bony prominences of the vertebrae and giving a quantitative score.
Adapted with permission from Wolfensohn & Honess 2005, please contact us for a fully accessible version if required.
-
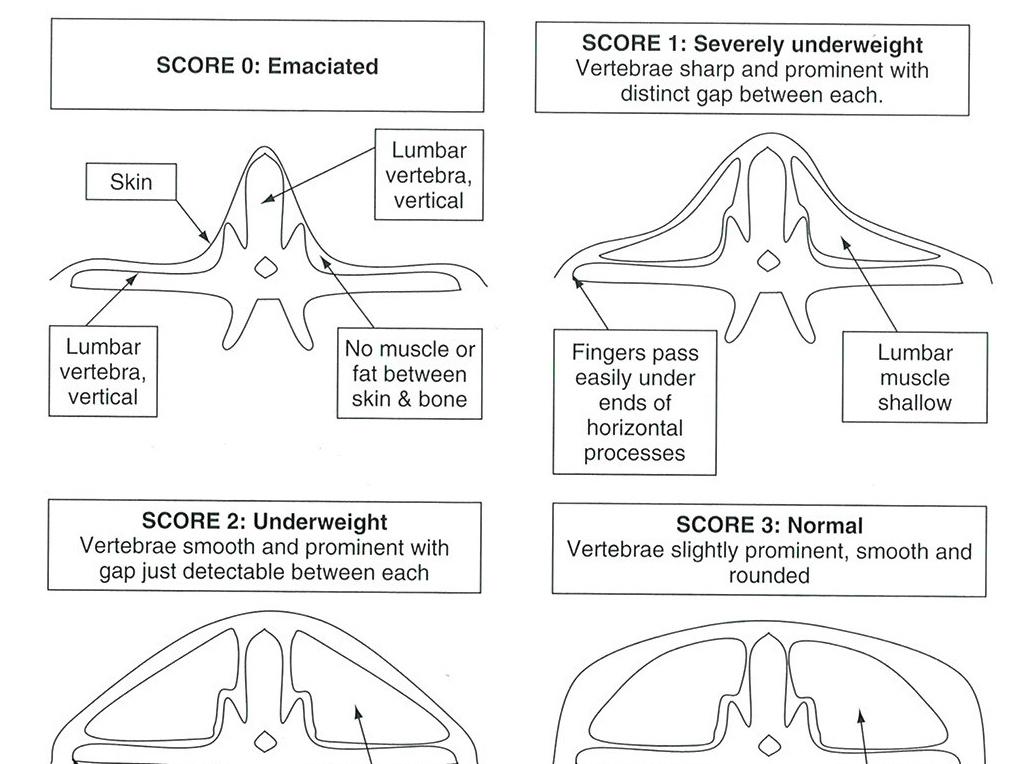 Body condition - Scoring by palpation - Wolfensohn & Honess.
Body condition - Scoring by palpation - Wolfensohn & Honess.
Hair loss can be common amongst captive primates, but is not so prevalent in wild animals [11], suggesting that some aspect of the captive environment contributes to abnormal patterns of loss [11]. It has a complex aetiology and is a multifactorial problem with many contributing factors [12,13]. Poor health, such as parasitic infections and skin diseases [14], nutrient specific deficient diets [15], hair pulling or over-grooming [16,17], stereotypical trichotillomania due to boredom and/or social stress [18] and reproductive conditions [19] have all been attributed as reasons for hair loss. The seasonal “molt” shown by rhesus macaques on Cayo Santiago is considered by some to be a natural, reversible phenomenon associated with reproductive seasonality [20,21].
Treatment via various behavioural methods (e.g. additional environmental enrichment) [13] does not always succeed in producing a reversal in severity [22], and therefore the link between hair loss and reduced welfare may be an oversimplification. Nonetheless, given its prevalence in captive animals, particularly in environments (social and physical) considered to be challenging to welfare, and that recipients of hair pulling often show fear or avoidance reactions [16] which are likely to reflect poor welfare [11], quantifying alopecia may be a useful tool for welfare assessment. Scoring involves estimating the severity and extent of hair loss according to a five point scoring system [11].
Signs of pain may include [2,23]:
- Sitting in a huddled position, or crouched with head forwards and down with arms across the body.
- Sad expression with glassy eyes.
- Reduced movement.
- Reduced appetite and/or reduced interest in food treats.
- Lack of grooming or poor coat.
- Avoiding conspecifics and/or staff.
- Increased attention from cage mates.
- Facial contortions, clenching of teeth.
- Restlessness and shaking, accompanied by vocalisation (e.g. grunts, groans).
- Touching, pushing or scratching the painful area; self mutilation.
- Increased or decreased aggressive toward caregivers (or other change in normal behaviour).
Behavioural signs such as these may not be specific to pain, and there is a need for more objective, rapid and reliable measures so that pain can be identified and alleviated more effectively. Researchers funded by the NC3Rs are validating the use of facial expressions for post-operative pain assessment in rhesus macaques, using a research tool established for analysing facial movement – the Macaque Facial Action Coding System. A grimace scale has been developed, similar to those for the mouse, rat and rabbit.
Examples of score sheets are given below.
In the case of numerical score sheets, a number of clinical signs, physical indicators and behavioural parameters are assessed and given a score according to their apparent severity. Often scores are added up and the results used to determine whether action is needed (such as analgesia) according to a predetermined key attached to the sheet. The animal is reassessed at appropriate intervals to monitor its progress and response to any treatments.
See Hawkins et al. 2011 for general advice on defining and implementing score sheets.
Examples of welfare assessment score sheets
- Objective measures of health and well-being in laboratory rhesus monkeys (Macaca mulatta)
Smith et al. (2006) have developed and validated an objective and quantitative system for assessing and monitoring the health and well-being of laboratory rhesus macaques; specifically those used in long-term neurophysiology studies with fluid control. Observations are made twice a day by an experienced observer and checklists used to record: (a) potentially life-threatening clinical concerns; (b) developing clinical issues; (c) atypical behaviours; and (d) laboratory performance. The authors demonstrate with two case studies the utility of their multidimensional system for identifying incipient clinical and behaviour problems before they become serious. Although observations were made twice daily, the authors comment that a single daily measure would be sufficient. - Handbook of primate husbandry and welfare
Wolfensohn & Honess 2005 present an example sheet for monitoring a monkey with a neurological deficit (p.62-63).
General welfare assessment score sheet
The welfare assessment score sheet below (adapted from Wolfensohn & Honess 2005) is presented to illustrate the general principles. It should be modified to provide specific information on the condition or research model under investigation – an accompanying excel sheet is available to download.
Download
-
Screenshot of the general welfare assessment spreadsheet available to download
References
- Summers L et al. (2012). Validation of a body condition scoring system in rhesus macaques (macaca mulatta): Assessment of body composition by using dual-energy x-ray absorptiometry. Journal of the American Association for Laboratory Animal Science : JAALAS 51(1): 88-93.
-
Wolfensohn S and Honess P (2005). Handbook of primate husbandry and welfare, 1st edition. Wiley-Blackwell.
-
Prescott MJ et al. (2010). Refinement of the use of food and fluid control as motivational tools for macaques used in behavioural neuroscience research: Report of a working group of the nc3rs. Journal of Neuroscience Methods 193(2): 167-88. https://doi.org/10.1016/j.jneumeth.2010.09.003
-
van Wagenen G and Catchpole HR (1956). Physical growth of the rhesus monkey (macaca mulatta). American Journal of Physical Anthropology 14(2): 245-73. https://doi.org/10.1002/ajpa.1330140219
-
Turnquist JE and Kessler MJ (1989). Free-ranging cayo santiago rhesus monkeys (macaca mulatta): I. Body size, proportion, and allometry. American Journal of Primatology 19(1): 1-13. https://doi.org/10.1002/ajp.1350190102
-
Schapiro SJ and Kessel AL (1993). Weight gain among juvenile rhesus macaques: A comparison of enriched and control groups. Laboratory animal science 43(4): 315-8
-
Andrade MCR et al. (2004). Biologic data of macaca mulatta, macaca fascicularis, and saimiri sciureusused for research at the fiocruz primate center. Memórias do Instituto Oswaldo Cruz 99(6): 584-9. https://doi.org/10.1590/S0074-02762004000600009
-
Wilk JL et al. (2008). Evaluation of the use of coconut to treat chronic diarrhea in rhesus macaques (macaca mulatta). Journal of Medical Primatology 37(6): 271-6. https://doi.org/10.1111/j.1600-0684.2008.00313.x
-
Prongay K et al. (2013). Risk factor analysis may provide clues to diarrhea prevention in outdoor-housed rhesus macaques (macaca mulatta). American Journal of Primatology 75(8): 872-82. https://doi.org/10.1002/ajp.22150
-
Kanthaswamy S et al. (2014). Familial aggregation of chronic diarrhea disease (cdd) in rhesus macaques (macaca mulatta). American Journal of Primatology 76(3): 262-70. https://doi.org/10.1002/ajp.22230
-
Honess PE et al. (2005). Alopecia scoring: The quantitative assessment of hair loss in captive macaques. Alternatives to Laboratory Animals 33(3): 193-206. https://doi.org/10.1177/026119290503300308
-
Beisner BA and Isbell LA (2009). Factors influencing hair loss among female captive rhesus macaques (macaca mulatta). Applied Animal Behaviour Science 119(1-2): 91-100. https://doi.org/10.1016/j.applanim.2009.03.016
-
Novak MA and Meyer JS (2009). Alopecia: Possible causes and treatments, particularly in captive nonhuman primates. Comparative medicine 59(1): 18-26.
-
Steinmetz HW et al. (2005). Dermatologic investigation of alopecia in rhesus macaques (macaca mulatta). Journal of Zoo and Wildlife Medicine 36(2): 229-38. https://doi.org/10.1638/04-054.1
-
Isbell LA (1995). Seasonal and social correlates of changes in hair, skin, and scrotal condition in vervet monkeys (cercopithecus aethiops) of amboseli national park, kenya. American Journal of Primatology 36(1): 61-70. https://doi.org/10.1002/ajp.1350360105
-
Reinhardt V et al. (1986). Hair pulling and eating in captive rhesus monkey troops. Folia Primatologica 47(2-3): 158-64. https://doi.org/10.1159/000156272
-
Reinhardt V (2005). Hair pulling: A review. Laboratory Animals 39(4): 361-9. https://doi.org/10.1258/002367705774286448
-
Steinmetz HW et al. (2006). Coat condition, housing condition and measurement of faecal cortisol metabolites - a non-invasive study about alopecia in captive rhesus macaques (macaca mulatta). Journal of Medical Primatology 35(1): 3-11. https://doi.org/10.1111/j.1600-0684.2005.00141.x
-
Davis EB and Suomi SJ (2006). Hair loss and replacement cycles in socially housed, pregnant, rhesus macaques. American Journal of Primatology 68(Supplementary): 1-58.
-
Vessey SH and Morrison JA (1970). Molt in free-ranging rhesus monkeys, macaca mulatta. Journal of Mammalogy 51(1): 89-93. https://doi.org/10.2307/1378535
-
Kessler MJ (2009). Alopecia: Possible causes and treatments, particularly in captive nonhuman primates. Comparative medicine 59(4): 320.
-
Kramer J et al. (2010). Alopecia in rhesus macaques correlates with immunophenotypic alterations in dermal inflammatory infiltrates consistent with hypersensitivity etiology. Journal of Medical Primatology 39(2): 112-22. https://doi.org/10.1111/j.1600-0684.2010.00402.x
-
International Primatological S (2007). Ips international guidelines for the acquisition, care and breeding of nonhuman primates, 2nd edition.