Being able to recognise, interpret and respond appropriately to their communication signals (such as facial expressions, postures and vocalisations) provides a good basis for refining many aspects of their care and use. The advice of a primate behaviour specialist is recommended for helping staff and animals to interact positively within the laboratory environment. For example ethograms and advice on recording behaviour, see the Behavioural indicators of welfare section.
Assess your knowledge and understanding of the behaviour of macaques, take our quiz on macaque behaviour.
A healthy captive environment allows macaques to spend their time engaged in a wide range of ‘natural’ behaviours. The activity budgets of wild macaques vary [1-5], but in all studies the macaques spend the majority of their time foraging, resting, allogrooming and moving around their environment. In captivity, animals that cannot spend their time in this way are at risk of becoming bored, depressed, frustrated, aggressive and/or stressed [6]. While daily patterns vary, wild macaques forage and travel most intensively in the morning and rest and allogroom throughout the afternoon.
-
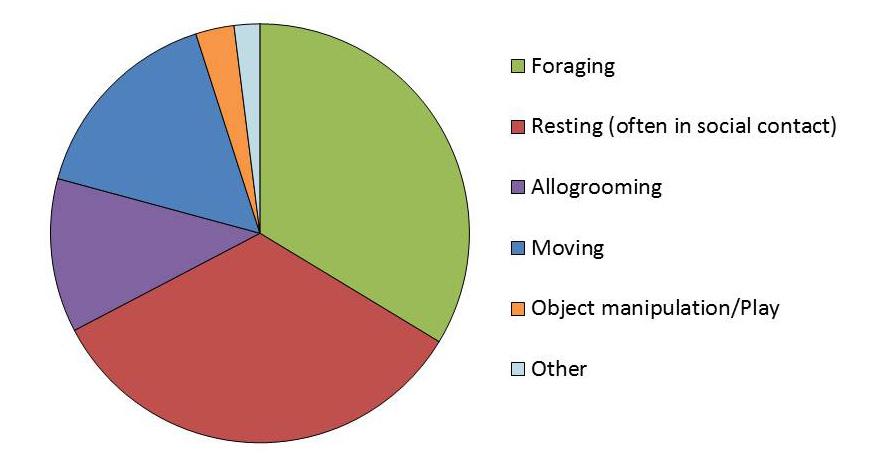 A pie chart showing rhesus macaque time budgets; they spend the majority of the time foraging and resting (often in social contact). Data for rhesus macaques, averaged from Jaman & Huffman 2013 and Zhou et al. 2013
A pie chart showing rhesus macaque time budgets; they spend the majority of the time foraging and resting (often in social contact). Data for rhesus macaques, averaged from Jaman & Huffman 2013 and Zhou et al. 2013
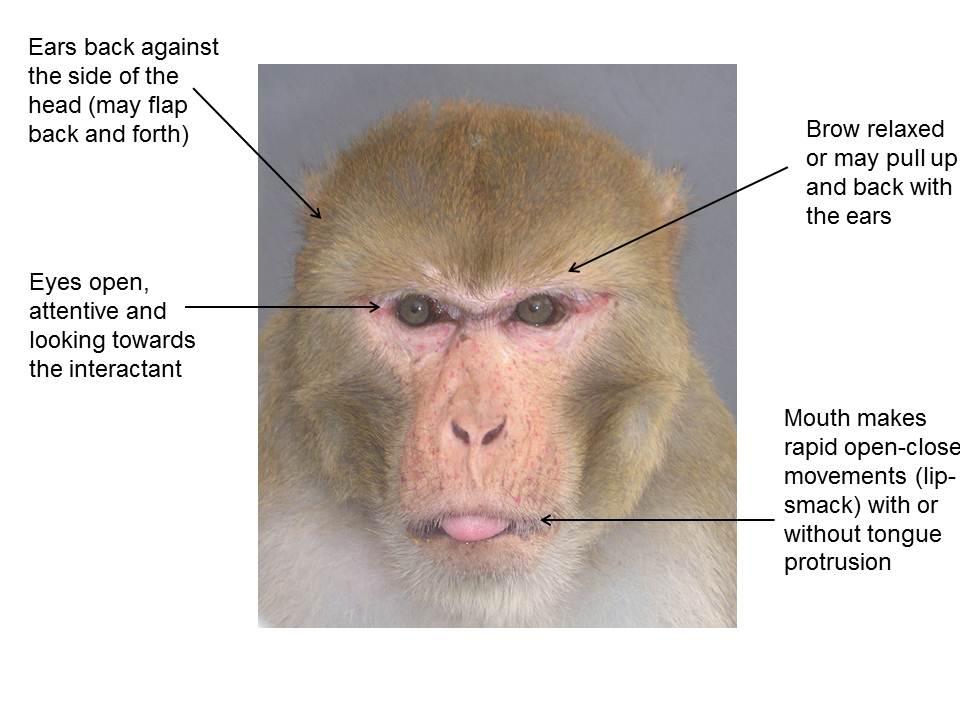
Macaques have a variety of facial expressions which they use to communicate with conspecifics, composed of movements of the ears, brow, eyes and mouth from the neutral state [7-9]. Facial expressions can be used by laboratory staff to infer the attention, intention and internal state of animals. However, the purpose of all macaque facial expressions is not yet fully understood. Expressions may be performed at different intensities in different contexts; there may be individual differences in how expressions appear; and an animal will often perform several expressions and behaviours together, as seen from the videos in this section. About one third of facial expressions are accompanied by vocalisation [9].
Information on using abnormal behaviours to assess welfare, and preventing abnormal behaviour, is given in the Behavioural indicators section.
Hair plucking is one sign of abnormal behaviour:
Macaques diets are highly varied:
- Rhesus macaques eat fruits, young and mature leaves, stems, seeds, flowers, petioles, bark and roots of over 75 different plant species [62].
- Cynomolgus macaques are predominantly frugivorous (fruit accounts for 67%-82% of their diet) [63,64], but also feed on other plant parts. Foraging is typically conducted below 20 m (65.6 ft); usually around 12 m (39.4 ft) in the lower canopy, understory, and on the ground [65]. At mangroves they have also been seen to consume bivalves, shrimp and octopus [66,67].
- Both species feed opportunistically on grass, clay, mushrooms, invertebrates, eggs, crabs, and small vertebrates such as lizards, frogs, birds and fish [68].
Non-exhaustive list of plant species and their component parts eaten by rhesus macaques
(Adapted from Zhou et al., 2014)
|
Plant: Latin name |
Plant: Common name |
Fruit eaten |
Stems eaten |
Young leaves eaten |
Mature leaves eaten |
|---|---|---|---|---|---|
|
Aristolochia longgangensis |
Dutchman’s pipe, Pipevine, Birthwort |
Yes |
Yes |
No |
No |
|
Capparis cahtohiesis |
– |
Yes |
No |
Yes |
Yes |
|
Carvota ochlandra |
Chinese fishtail palm |
Yes |
No |
Yes |
No |
|
Clausena anisum |
Anis (Philippines) |
Yes |
No |
No |
No |
|
Clausena emarginata |
Powderpuff plant, Cat’s tail |
Yes |
No |
Yes |
Yes |
|
Cuscuta chinensis |
Cuscuta, Chinese dodder |
No |
Yes |
No |
No |
|
Dracontomelon duperreanum |
Yanmin |
Yes |
No |
Yes |
No |
|
Embelia scandens |
Mez |
No |
No |
Yes |
Yes |
|
Ficus gibbosa |
Fig |
Yes |
No |
Yes |
Yes |
|
Ficus glaberrima |
Fig |
Yes |
No |
No |
Yes |
|
Ficus macrocarpa |
Curtain fig, Chinese banyan, Indian laurel |
Yes |
No |
Yes |
Yes |
|
Ficus obscura |
Fig, Appolas |
Yes |
No |
No |
No |
|
Indocalamus calcicolus |
Bamboo |
No |
No |
Yes |
No |
|
Iondes ovalis |
– |
Yes |
No |
Yes |
Yes |
|
Pithecellobium clypearia |
Greater grasshopper tree |
No |
No |
Yes |
Yes |
|
Pothos repens |
– |
Yes |
No |
Yes |
No |
|
Polygonum chinense |
Chinese knotweed |
Yes |
No |
No |
No |
|
Pueraria thunbergiana |
Kudzu, Japanese arrowroot |
No |
Yes |
Yes |
Yes |
|
Spondia lakonensis |
– |
Yes |
No |
Yes |
No |
|
Urobotrya latisquama |
Hiepko |
Yes |
No |
Yes |
Yes |
Diet composition of cynomolgus macaques
(Adapted from Brotcorne 2014)
|
Location |
Origin of food (%) |
Fruit (%) |
Leaves (%) |
Flowers |
Animal matter (%) |
Other (%) |
|---|---|---|---|---|---|---|
|
Malaysia |
Natural |
52.4 |
19 |
5.4 |
23.2 |
0 |
|
Singapore |
Natural (86) and anthropogenic (14) |
44 |
8 |
7 |
0 |
41 |
|
Singapore |
Natural (51) and anthropogenic (49) |
20.9 |
21 |
6.3 |
0 |
51.8 |
|
Singapore
|
Natural (74) and anthropogenic (26) |
44.8 |
19.6 |
9.7 |
0 |
28.3 |
|
Kalimantan. Borneo |
Natural |
66.7 |
17.2 |
8.9 |
4.1 |
3.1 |
|
Kalimantan. Borneo |
Natural |
87 |
4 |
3 |
4 |
2 |
|
Ubud, Bali |
Natural (48) and anthropogenic (52) |
32 |
34 |
5 |
29 |
0 |
|
Ubud, Bali |
Natural (30) and anthropogenic (70) |
25.7 |
14.7 |
0 |
7.5 |
52.1 |
|
TNBB, Bali |
Natural (89) and anthropogenic (11) |
60 |
21 |
0 |
3 |
16 |
|
Uluwatu, Bali |
Natural (52) and anthropogenic (48) |
32 |
19 |
0 |
14 |
35 |
|
Vietnam |
Natural (75) and anthropogenic (25) |
15 |
20 |
1 |
35 |
29 |
|
Mauritius |
Natural and anthropogenic |
42.2 |
30.8 |
7 |
2.4 |
17.6 |
References
- Jaman MF and Huffman MA (2013). The effect of urban and rural habitats and resource type on activity budgets of commensal rhesus macaques (macaca mulatta) in bangladesh. Primates 54(1): 49-59. https://doi.org/10.1007/s10329-012-0330-6
- Zhou Q et al. (2014). Niche separation of sympatric macaques, macaca assamensis and m. Mulatta, in limestone habitats of nonggang, china. Primates 55(1): 125-37. https://doi.org/10.1007/s10329-013-0385-z
-
Li D et al. (2012). Sex-age differences in activity budget and position behavior of rhesus macaques (macaca mulatta). Acta Theriologica Sinica 32(1): 25-32.
-
Hambali K et al. (2012). Daily activity budget of long-tailed macaques (macaca fascicularis) in kuala selangor nature park. International Journal of Basic & Applied Sciences 12(4): 47-52.
-
Majolo B et al. (2013). The effect of climatic factors on the activity budgets of barbary macaques (macaca sylvanus). International Journal of Primatology 34(3): 500-14. https://doi.org/10.1007/s10764-013-9678-8
-
Mallapur A et al. (2005). Factors influencing the behaviour and welfare of captive lion-tailed macaques in indian zoos. Applied Animal Behaviour Science 91(3-4): 337-53. https://doi.org/10.1016/j.applanim.2004.10.002
-
Hinde RA and Rowell TE (1962). Communication by postures and facial expressions in the rhesus monkey (macaca mulatta). Proceedings of the Zoological Society of London 138(1): 1-21. https://doi.org/10.1111/j.1469-7998.1962.tb05684.x
-
Parr LA et al. (2010). Brief communication: Maqfacs: A muscle-based facial movement coding system for the rhesus macaque. American Journal of Physical Anthropology 143(4): 625-30. https://doi.org/10.1002/ajpa.21401
-
Partan SR (2002). Single and multichannel signal composition: Facial expressions and vocalizations of rhesus macaques (macaca mulatta). Source: Behaviour 139(8): 993-1027.
-
Rowe N (1996). The pictorial guide to the living primates. Pogonias Pr.
-
Maestripieri D (1999). Primate social organization, gestural repertoire size, and communication dynamics: A comparative study of macaques. In: The evolution of language: Assessing the evidence from nonhuman primates (Ed., King BJ) (pp. 55-77). School of American Research.
-
Maestripieri D (2005). Gestural communication in three species of macaques (macaca mulatta , m. Nemestrina , m. Arctoides). Gesture 5(1-2): 57-73. https://doi.org/10.1075/gest.5.1.06mae
-
Moynihan M (1976). The new world primates: Adaptive rediation and the evolution of social behavior, languages, and intelligence. Princeton University Press.
-
Menzel CR (1980). Head-cocking and visual perception in primates. Animal Behaviour 28(1): 151-IN10. https://doi.org/10.1016/S0003-3472(80)80020-0
-
Cantalupo C et al. (2001). Function of head-cocking in garnett’s greater bush baby (otolemur garnettii). International Journal of Primatology 11(1).
-
Hauser MD (1998). Functional referents and acoustic similarity: Field playback experiments with rhesus monkeys. Anim Behav 55: 1647-58.
-
Whitham JC et al. (2007). Intended receivers and functional significance of grunt and girney vocalizations in free-ranging female rhesus macaques. Ethology 113(9): 862-74. https://doi.org/10.1111/j.1439-0310.2007.01381.x
-
Lindburg DG (1971). The rhesus monkey in north india : An ecological and behavioral study. In: Primate behavior: Developments in field and laboratory research (Ed., Rosenblum LA) (Vol. 2, pp. 1-106). Academic Pr.
-
Palombit RA (1992). A preliminary study of vocal communication in wild long-tailed macaques (macaca fasc/adar/s). I. Vocal repertoire and call emission. International Journal of Primatology 13.
-
Wheatley BP (1999). The sacred monkeys of bali, 1st edition. Waveland Press.
-
Lindburg DG (1971). The rhesus monkey in north india : An ecological and behavioral study. In: Primate behavior: Developments in field and laboratory research (Ed., Rosenblum LA) (Vol. 2, pp. 1-106). Academic Pr.
-
Maestripieri D and Roney JR (2005). Primate copulation calls and postcopulatory female choice. Behavioral Ecology 16(1): 106-13. https://doi.org/10.1093/beheco/arh120
-
Raffles TS (1821). Xvii. Descriptive catalogue of a zoological collection, made on account of the honourable east india company, in the island of sumatra and its vicinity, under the direction of sir thomas stamford raffles, lieutenant-governor of fort marlborough; with addi. Transactions of the Linnean Society of London 13(1): 239-74. https://doi.org/10.1111/j.1095-8339.1821.tb00064.x
-
Rowell T (1962). Agonistic noises of the rhesus monkey (macaca mulatta). Sym Zool Soc London 8: 91-6.
-
Gouzoules S et al. (1984). Rhesus monkey (macaca mulatta) screams: Representational signalling in the recruitment of agonistic aid. Animal Behaviour 32(1): 182-93. https://doi.org/10.1016/S0003-3472(84)80336-X
-
van Noordwijk MA (2010). Sexual behaviour of sumatran long-tailed macaques (macaca fascicularis). Zeitschrift für Tierpsychologie 70(4): 277-96. https://doi.org/10.1111/j.1439-0310.1985.tb00519.x
-
Augustsson H and Hau J (1999). A simple ethological monitoring system to assess social stress in group-housed laboratory rhesus macaques. Journal of Medical Primatology 28(2): 84-90. https://doi.org/10.1111/j.1600-0684.1999.tb00255.x
-
Massen JJM and Sterck EHM (2013). Stability and durability of intra- and intersex social bonds of captive rhesus macaques (macaca mulatta). International Journal of Primatology 34(4): 770-91. https://doi.org/10.1007/s10764-013-9695-7
-
Rakhovskaya MV (2013). Correlates of male consortship rate in free-ranging rhesus macaques (macaca mulatta). International Journal of Primatology 34(4): 662-80. https://doi.org/10.1007/s10764-013-9686-8
-
Gumert MD (2007). Grooming and infant handling interchange in macaca fascicularis: The relationship between infant supply and grooming payment. International Journal of Primatology 28(5): 1059-74. https://doi.org/10.1007/s10764-007-9202-0
-
Cords M (1992). Post-conflict reunions and reconciliation in long-tailed macaques. Animal Behaviour 44(1): 57-61. https://doi.org/10.1016/S0003-3472(05)80754-7
-
Aureli F and Schaik CPv (2010). Post-conflict behaviour in long-tailed macaques (macaca fascicularis). Ethology 89(2): 89-100. https://doi.org/10.1111/j.1439-0310.1991.tb00296.x
-
Demaria C and Thierry B (2001). A comparative study of reconciliation in rhesus and tonkean macaques. Behaviour 138(3): 397-410. https://doi.org/10.1163/15685390152032514
-
Call J et al. (1999). Reconciliation patterns among stumptailed macaques: A multivariate approach. Animal Behaviour 58(1): 165-72. https://doi.org/10.1006/anbe.1999.1116
-
Russell YI and Phelps S (2013). How do you measure pleasure? A discussion about intrinsic costs and benefits in primate allogrooming. In Biology and Philosophy (Vol. 28, pp. 1005-20).
-
Dunbar RIM (2010). The social role of touch in humans and primates: Behavioural function and neurobiological mechanisms. Neuroscience & Biobehavioral Reviews 34(2): 260-8. https://doi.org/10.1016/j.neubiorev.2008.07.001
-
Shutt K et al. (2007). Grooming in barbary macaques: Better to give than to receive? Biology Letters 3(3): 231-3. https://doi.org/10.1098/rsbl.2007.0052
-
Simpson EA et al. (2014). Inhaled oxytocin increases positive social behaviors in newborn macaques. Proceedings of the National Academy of Sciences 111(19): 6922-7. https://doi.org/10.1073/pnas.1402471111
-
Winslow JT et al. (2003). Rearing effects on cerebrospinal fluid oxytocin concentration and social buffering in rhesus monkeys. Neuropsychopharmacology 28(5): 910-8. https://doi.org/10.1038/sj.npp.1300128
-
Chang SWC et al. (2012). Inhaled oxytocin amplifies both vicarious reinforcement and self reinforcement in rhesus macaques (macaca mulatta). Proceedings of the National Academy of Sciences 109(3): 959-64. https://doi.org/10.1073/pnas.1114621109
-
Higham JP et al. (2011). Mu-opioid receptor (oprm1) variation, oxytocin levels and maternal attachment in free-ranging rhesus macaques macaca mulatta. Behavioral Neuroscience 125(2): 131-6. https://doi.org/10.1037/a0022695
-
Hinde RA and Spencer-Booth Y (1967). The behaviour of socially living rhesus monkeys in their first two and a half years. Animal Behaviour 15(1): 169-96. https://doi.org/10.1016/S0003-3472(67)80029-0
-
Brown GR and Dixson AE (2000). The development of behavioural sex differences in infant rhesus macaques (macaca mulatta). Primates 41(1): 63-.
-
Hassett JM et al. (2009). Social segregation in male, but not female yearling rhesus macaques (macaca mulatta). American Journal of Primatology:. https://doi.org/10.1002/ajp.20756
-
Prescott MJ et al. (2012). Laboratory macaques: When to wean? Applied Animal Behaviour Science 137(3-4): 194-207. https://doi.org/10.1016/j.applanim.2011.11.001
-
Estes RD (1991). The behavior guide to African mammals. University of California Press.
-
Wallen K et al. (1984). Periovulatory changes in female sexual behavior and patterns of ovarian steroid secretion in group-living rhesus monkeys. Hormones and Behavior 18(4): 431-50. https://doi.org/10.1016/0018-506X(84)90028-X
-
Rowell TE (1963). Behaviour and female reproductive cycles of rhesus macaques. Reproduction 6(2): 193-203. https://doi.org/10.1530/jrf.0.0060193
-
Dunk RDP (2013). Seasonality of conceptions under varying conditions in a rhesus macaque breeding colony. Thesis. University of Wisconsin-Milwaukee
-
Hambali K et al. (2012). Daily activity budget of long-tailed macaques (macaca fascicularis) in kuala selangor nature park. International Journal of Basic & Applied Sciences 12(4): 47-52.
-
Fooden J (2000). Systematic review of the rhesus macaque, macaca mulatta (zimmermann, 1780) / jack fooden 1 ed. Field Museum of Natural History. https://doi.org/10.5962/bhl.title.7192
-
Xu F et al. (2012). Construction and Validation of a Systematic Ethogram of Macaca fascicularis in a Free Enclosure. PLOS ONE 7(5): e37486. doi:10.1371/journal.pone.0037486
-
Jaman MF and Huffman MA (2013). The effect of urban and rural habitats and resource type on activity budgets of commensal rhesus macaques (macaca mulatta) in bangladesh. Primates 54(1): 49-59. https://doi.org/10.1007/s10329-012-0330-6
-
Dunbar DC (1989). Locomotor behavior of rhesus macaques (macaca mulatta) on cayo santiago. Puerto Rico health sciences journal 8(1): 79-85.
-
Dunbar DC (1989). Locomotor behavior of rhesus macaques (macaca mulatta) on cayo santiago. Puerto Rico health sciences journal 8(1): 79-85.
-
Riopelle AJ and Hubbard DG (1982). Vestibular function and development of swimming behavior in rhesus monkeys. Journal of Human Evolution 11(5): 377-82. https://doi.org/10.1016/S0047-2484(82)80092-4
-
Nahallage CAD and Huffman MA (2012). Macaque–human interactions in past and present-day sri lanka. In: The macaque connection. Developments in primatology: Progress and prospects (Eds. Radhakrishna S, Huffman MA, and Sinha A) (Vol. 43, pp. 135-48). Springer.
-
Nahallage CAD and Huffman MA (2008). Comparison of stone handling behavior in two macaque species: Implications for the role of phylogeny and environment in primate cultural variation. American Journal of Primatology 70(12): 1124-32. https://doi.org/10.1002/ajp.20610
-
Gumert MD et al. (2009). The physical characteristics and usage patterns of stone axe and pounding hammers used by long-tailed macaques in the andaman sea region of thailand. American Journal of Primatology 71(7): 594-608. https://doi.org/10.1002/ajp.20694
-
Wheatley BP (1980). Feeding and ranging of east bornean macaca fascicularis. In: The macaques: Studies in ecology, behavior, and evolution (Ed., Lindburg DG) (pp. 215-46). Van Nostrand Reinhold.
-
Erwin J and Deni R (1979). Strangers in a strange land: Abnormal behaviors or abnormal environments? In: Captivity and behavior: Primates in breeding colonies, laboratories and zoos (Eds. Maple TL, Erwin J, and Mitchell G) (pp. 1-28). Van Nostrand Reinhold.
-
Zhou Q et al. (2014). Niche separation of sympatric macaques, macaca assamensis and m. Mulatta, in limestone habitats of nonggang, china. Primates 55(1): 125-37. https://doi.org/10.1007/s10329-013-0385-z
-
Wich SA (2002). Measuring fruit patch size for three sympatric indonesian primate species. Primates 43: 19-27.
-
Ungar PS (1996). Feeding height and niche separation in sympatric sumatran monkeys and apes. Folia Primatologica 67(3): 163-8. https://doi.org/10.1159/000157218
-
Sussman RW and Tattersall I (1986). Distribution, abundance, and putative ecological strategy of macaca fascicularis on the island of mauritius, southwestern indian ocean. Folia Primatologica 46(1): 28-43. https://doi.org/10.1159/000156234
-
Son VD (2003). Diet of macaca fascicularis in a mangrove forest, vietnam. Laboratory Primate Newsletter 42(4): 1-5.
-
Wheatley BP (1980). Feeding and ranging of east bornean macaca fascicularis. In: The macaques: Studies in ecology, behavior, and evolution (Ed., Lindburg DG) (pp. 215-46). Van Nostrand Reinhold.























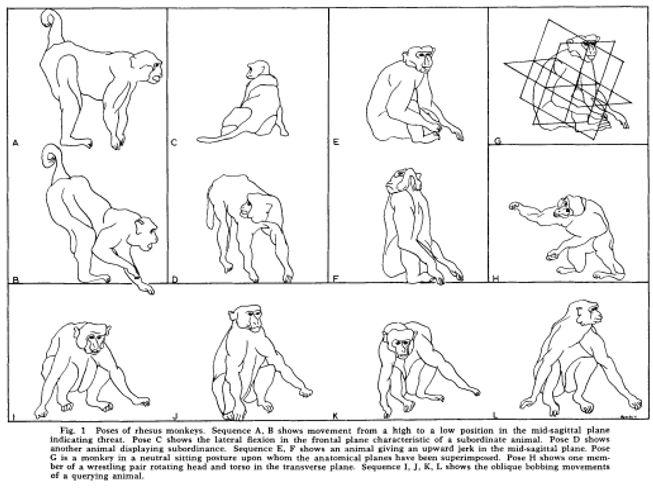






Social
In this section
Affiliative
Allo-grooming
One animal picks through the hair of another with hands or teeth, removing skin, dirt or ticks [7]. Allo-grooming is used to develop and maintain bonds between individuals. Females form stable life-long attachments, maintained through grooming, touch and close proximity [28]. Males groom females more during the mating season [29,30]. Macaques may groom after conflicts to console each other or repair damaged relationships [31-34]. Both giving and receiving grooming releases B-endorphins; after conflict situations, these may reduce pain from injury and relieve stress more quickly [35-37].
Affiliative contact
Macaques often rest and sleep huddled in family groups; they will also huddle when under threat. In behavioural studies, affiliation is often measured as proximity (e.g. within an arm’s reach of each other), because macaques that are friendly with each other will remain close, to maintain bonds and provide assistance if needed. The neuromodulators oxytocin and serotonin are implicated in development and maintenance of social behaviours such as approach and close contact [38-41]. These contribute to a sense of security from being close to conspecifics and have health benefits, such as maintaining low heart rate.
Present for grooming
An animal will approach or reposition itself in front of another, presenting an area of the body to be groomed. Typically seen during peaceful contexts, or by individuals seeking comfort after a fight.
Play
Mostly seen in infants, juveniles and adolescents. Play develops social bonds and motor-coordination skills, allowing younger animals to develop and assess their relative strength to peers. There are sex differences in play: females engage in more play with infants and older females; males engage in more rough-and-tumble play [42-44]. Adult males may engage older juveniles in play [20].
Infant care and handling
Infant care is performed mainly by nursing mothers during the birthing season. However, other group members are very interested in new infants. Looking after the infants of higher ranking females, either by a low-ranking female or male, will help improve their standing with the mother. The mother-infant bond in macaques is strong, and mediated by oxytocin released during birth and lactation throughout the first year of life [41]. It is important that infants are not removed from the mother before natural weaning has occurred, as this will lead to social attachment problems later in life [45].
Sexual
Genital present
When in oestrous and proceptive, a female will present her rump to the male to solicit copulation [46]. Outside of mating, both males and females may present their rump as a signal of subordination [47].
Mount and copulate
Reproductive behaviours are triggered by seasonal changes in sex hormones (e.g. oestrogen, testosterone). These may be accompanied by reddening of sexual skin on the face, genitals and rump. Mating is highly seasonal in the wild [48,49]. In captivity, mating seasons may be less pronounced.
Agonistic and aggressive
Displacement
Displacement most often occurs during competition for valued resources such as food, mates, shelter and good resting places. The dominant animal approaches a subordinate, who then moves away to avoid potential conflict. Dominant animals exert their dominance in subtle ways, which helps to conserve energy. In captivity, distributing resources so that they cannot be monopolised by dominant animals reduces stress for subordinates.
Aggressive approach
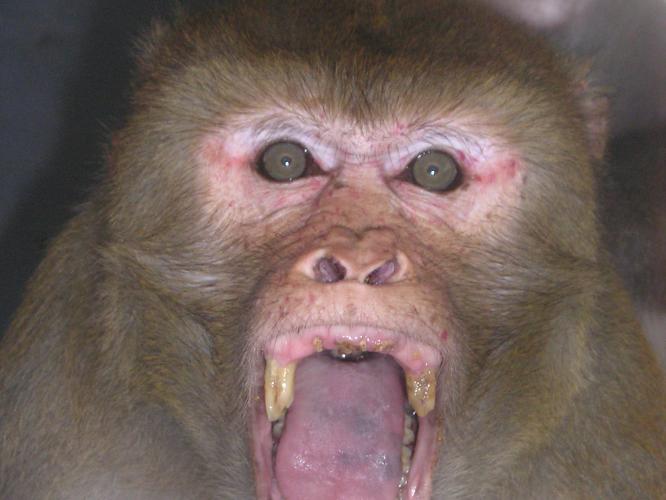
Approaching with staring face and an aggressive stance, including lunging with the tail up, are signs of dominance. Aggressive approach is typically performed by a dominant animal towards a subordinate.
Chase
Dominant animals will chase lower ranked animals away from resources where there is high competition, and use aggression to gain access.
Slap ground
Threat action, often accompanied by open mouth stare. For videos of slap ground, see the RHVIDEO site.
Lunge
Threat action
Branch shake
Size and aggression display to drive away potential threats. Most frequently given by males in response to other males, humans or perceived threats. In captivity, macaques will sometimes shake the home cage structure [7].
Dominant mount
An animal may exert its dominance over another by mounting it. Dominant mounting often occurs during tense situations, as dominant animals seek to re-affirm the hierarchy.
Push, Grab, Fight/Wrestle, Bite, Hit
Physical contact behaviours which can result in injury only occur when aggressive interactions cannot settled by non-contact means.
Stare
Threat action
Submissive and fearful
Avoidance
Subordinates will avoid or move away from dominant or aggressive animals, to avoid physical contact which may result in injury.
Rump present
Rump present indicates submission, performed by subordinates towards dominants [11]. Females may also present their rumps to males to solicit copulation.
Lean away
During potentially threatening social situations, macaques may lean away from a conspecific to avoid physical contact which may result in injury. For video of lean away, see the RHVIDEO site.
Look away
A subordinate will turn its back to a dominant individual to avoid eye contact and potential aggression.
Flee
Flee is a fear response in which a macaque runs away from a perceived threat. Seen during aggressive encounters, in the presence of predators, and when surprised [7].
Freeze
Freeze is a response in which a macaque stops all activity and remains still while assessing threat. Seen during aggressive encounters, in the presence of predators, and when surprised.